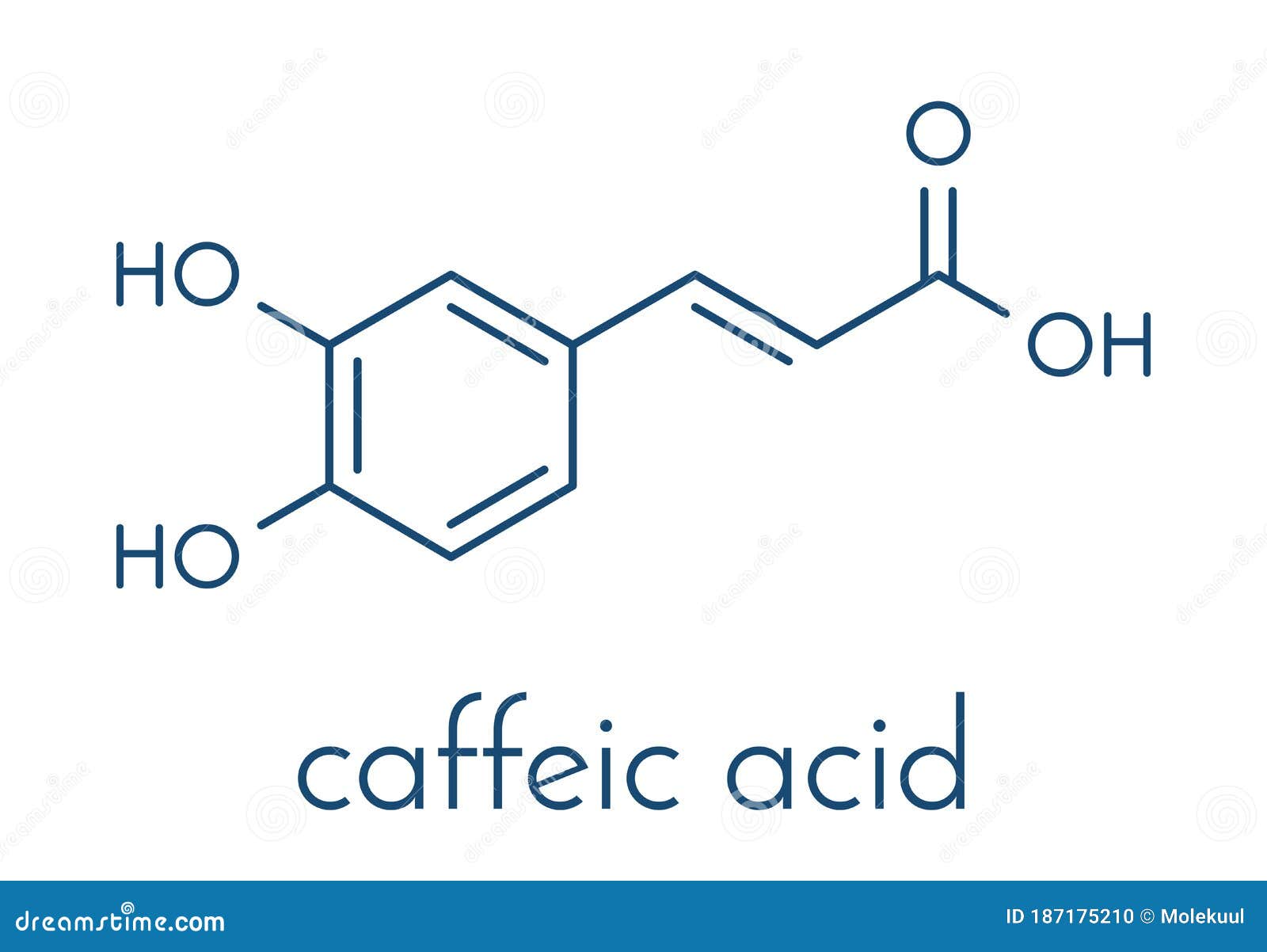
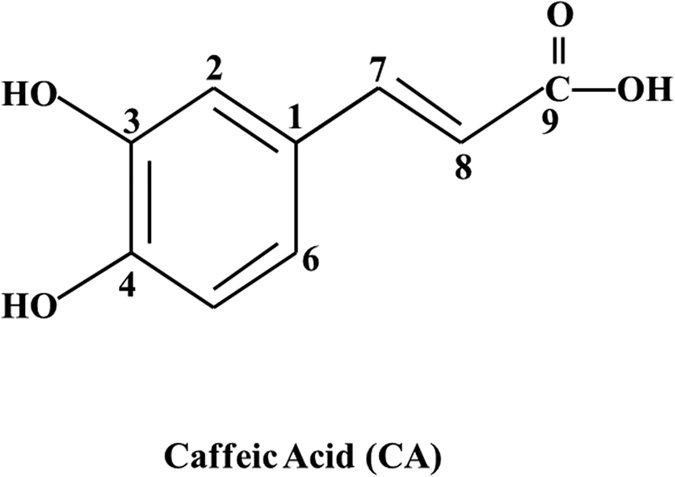
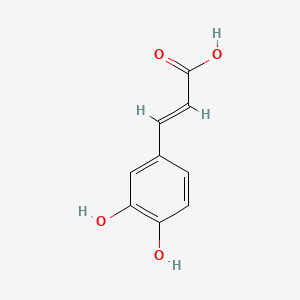
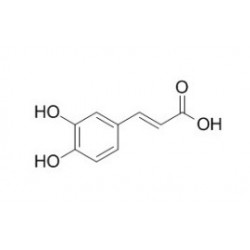
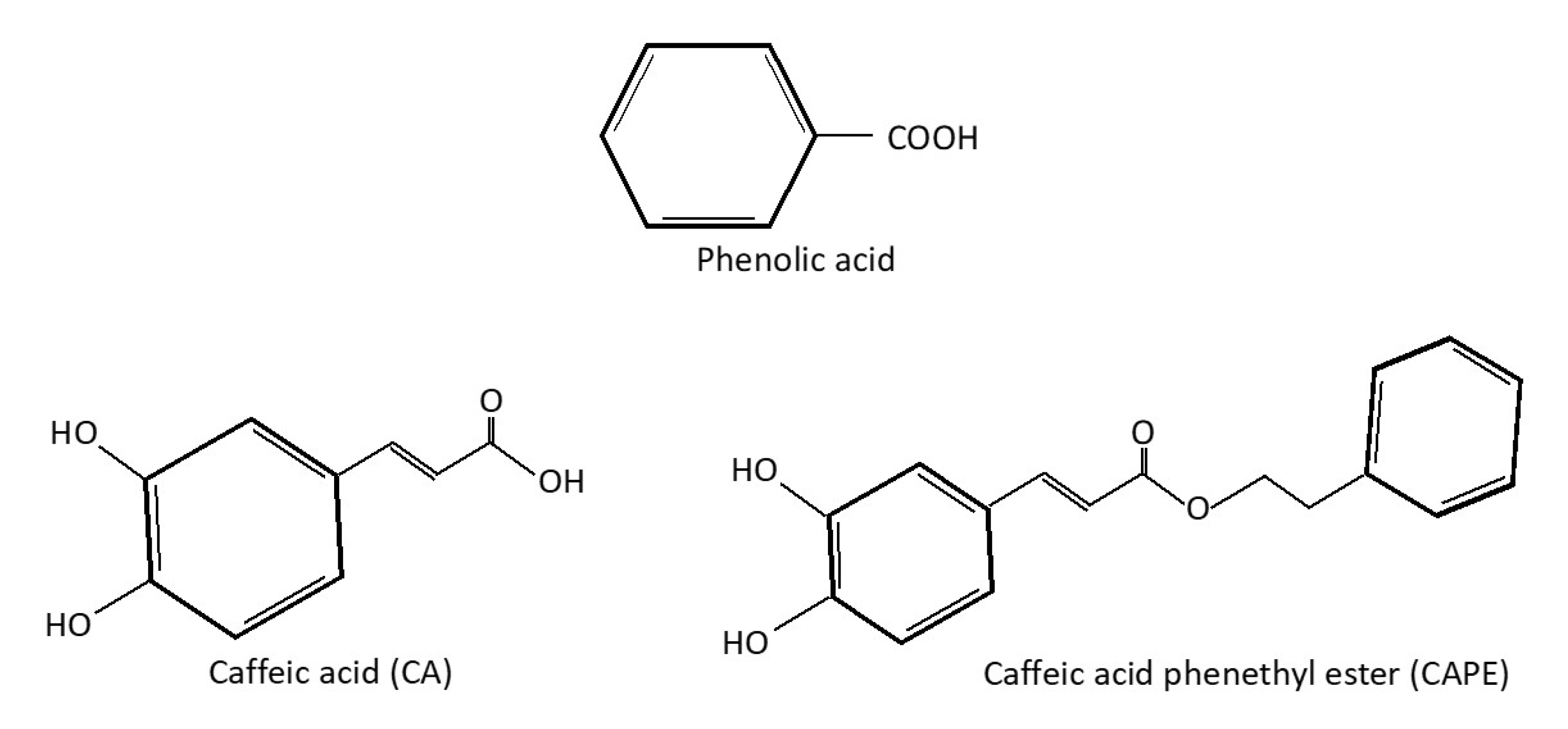
On the Inhibition of Hydroxyl Radical Formation by Hydroxycinnamic Acids: The Case of Caffeic Acid as a Promising Chelating Ligand of a Ferrous Ion | The Journal of Physical Chemistry A

Synthesis and evaluation of a series of caffeic acid derivatives as anticancer agents - ScienceDirect

Caffeic Acid and Its Derivatives: Antimicrobial Drugs toward Microbial Pathogens | Journal of Agricultural and Food Chemistry
![Caffeic acid N-[3,5-bis(trifluoromethyl)phenyl] amide as a non-steroidal inhibitor for steroid 5α-reductase type 1 using a human keratinocyte cell-based assay and molecular dynamics | Scientific Reports Caffeic acid N-[3,5-bis(trifluoromethyl)phenyl] amide as a non-steroidal inhibitor for steroid 5α-reductase type 1 using a human keratinocyte cell-based assay and molecular dynamics | Scientific Reports](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41598-022-25335-7/MediaObjects/41598_2022_25335_Fig1_HTML.png)
Caffeic acid N-[3,5-bis(trifluoromethyl)phenyl] amide as a non-steroidal inhibitor for steroid 5α-reductase type 1 using a human keratinocyte cell-based assay and molecular dynamics | Scientific Reports
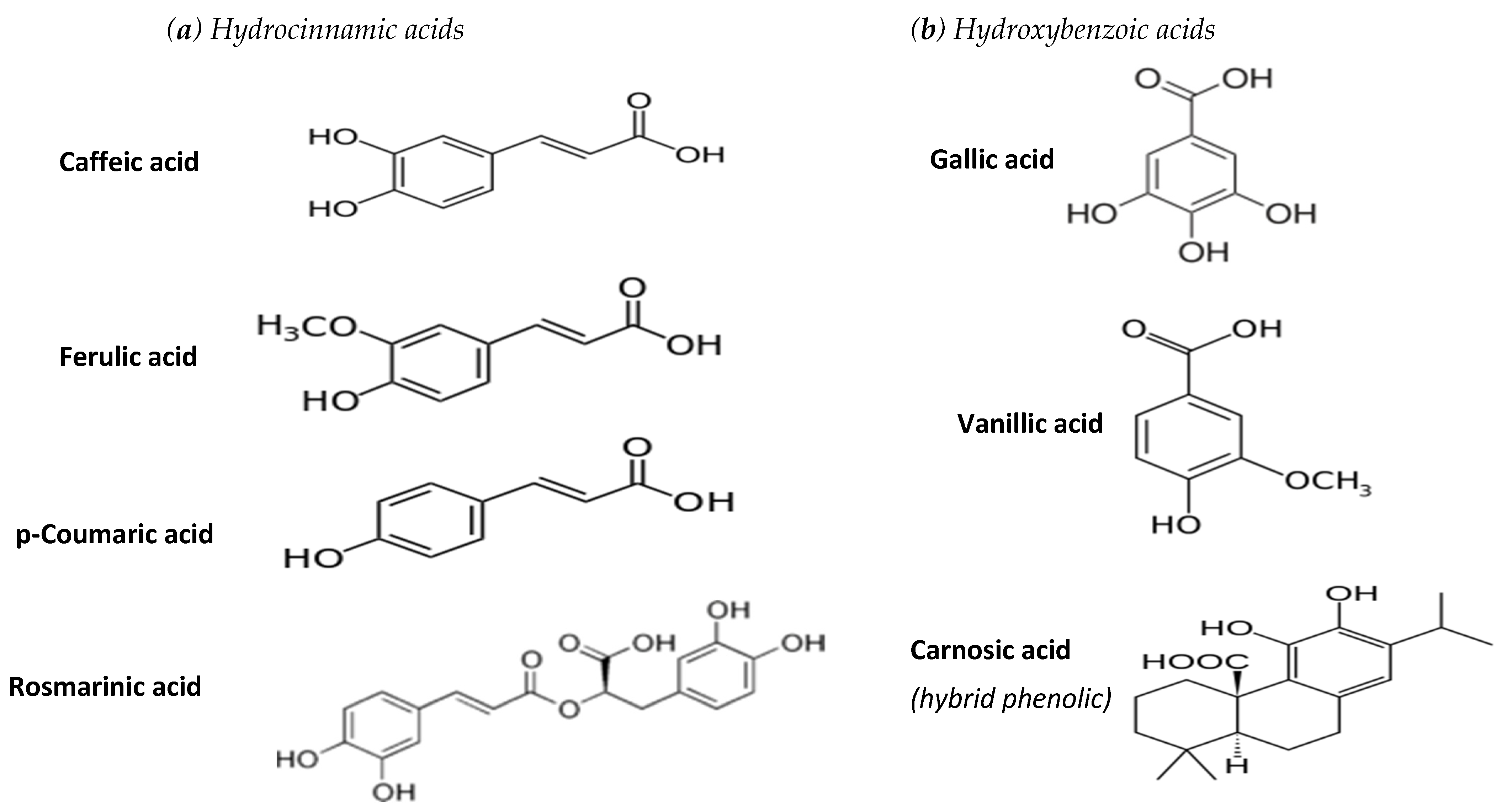
Molecules | Free Full-Text | A Review of the Health Protective Effects of Phenolic Acids against a Range of Severe Pathologic Conditions (Including Coronavirus-Based Infections)

Chemical and Pharmacological Aspects of Caffeic Acid and Its Activity in Hepatocarcinoma | Semantic Scholar