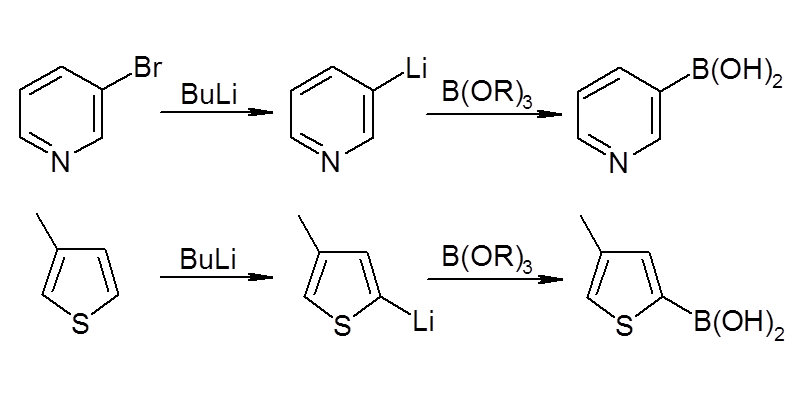
A Modular Synthesis of Teraryl‐Based α‐Helix Mimetics, Part 2: Synthesis of 5‐Pyridine Boronic Acid Pinacol Ester Building Blocks with Amino Acid Side Chains in 3‐Position - Peters - 2013 - Chemistry –
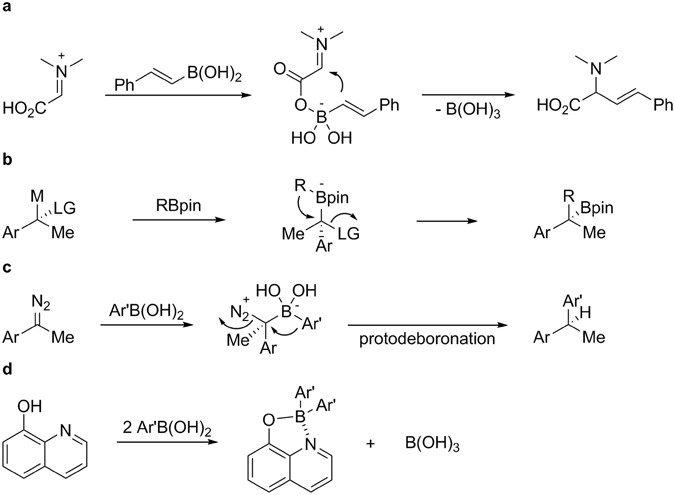
One-pot synthesis of four-coordinate boron(III) complexes by the ligand-promoted organic group migration between boronic acids | Scientific Reports

WO2013016185A1 - Synthesis of boronic esters and boronic acids using grignard reagents - Google Patents

WO2013016185A1 - Synthesis of boronic esters and boronic acids using grignard reagents - Google Patents

Molecules | Free Full-Text | Boronic Acids and Their Derivatives in Medicinal Chemistry: Synthesis and Biological Applications
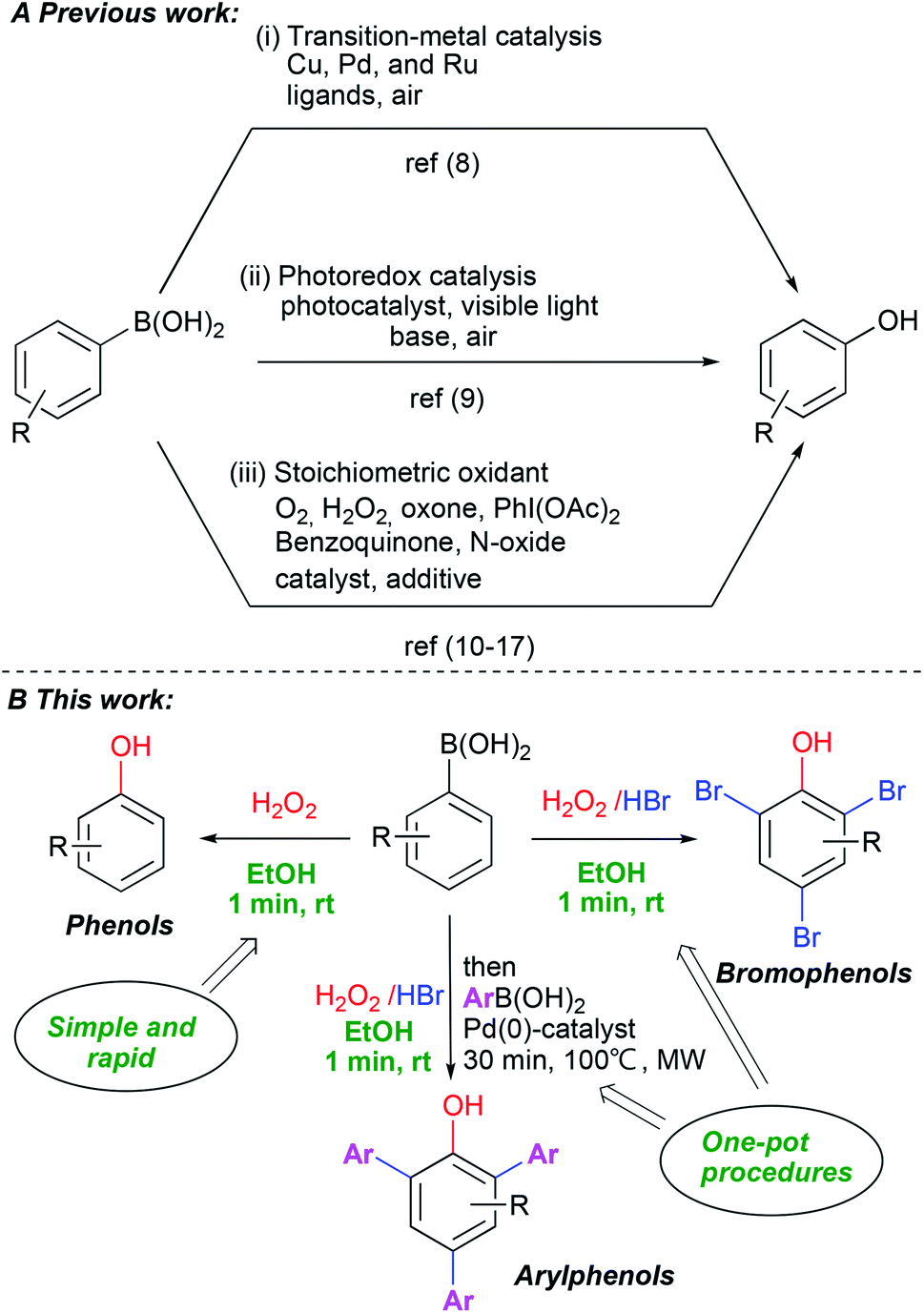
A scalable and green one-minute synthesis of substituted phenols - RSC Advances (RSC Publishing) DOI:10.1039/D0RA08580D

Enantiopure β-isocyano-boronic esters: synthesis and exploitation in isocyanide-based multicomponent reactions | SpringerLink




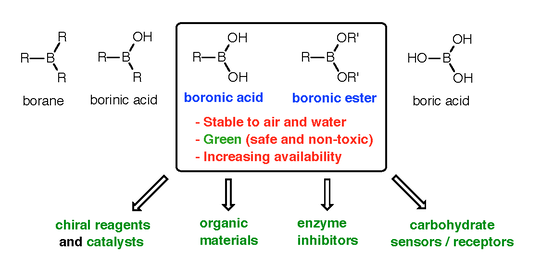
![PDF] Synthesis and Application of Boronic Acid Derivatives | Semantic Scholar PDF] Synthesis and Application of Boronic Acid Derivatives | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cb9ab1d15bd2639c72db5972e6edc06b85e60ae3/13-Figure1.2-1.png)